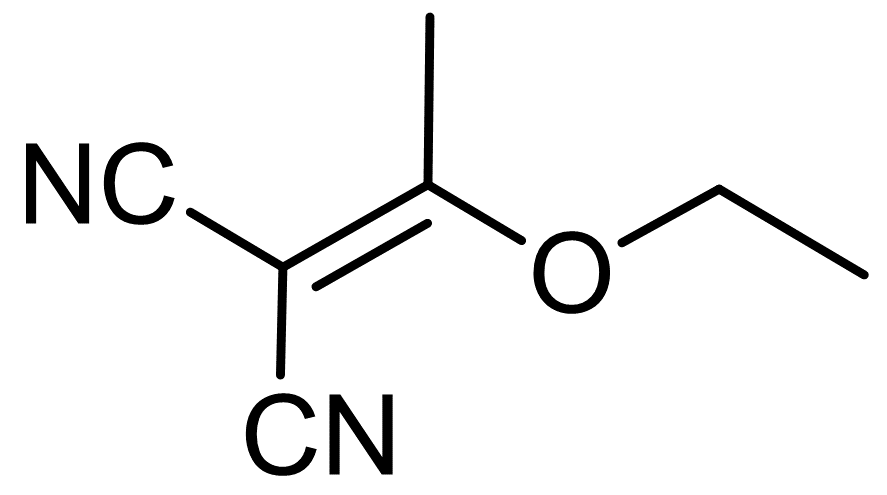
Propanedinitrile, (1-ethoxyethylidene)-
ethoxyethylidene malononitrile
CAS: 5417-82-3
Molecular Formula: C7H8N2O
Propanedinitrile, (1-ethoxyethylidene)- - Names and Identifiers
Propanedinitrile, (1-ethoxyethylidene)- - Physico-chemical Properties
| Molecular Formula | C7H8N2O |
| Molar Mass | 136.15 |
| Density | 1.1778 (rough estimate) |
| Melting Point | 90-92°C(lit.) |
| Boling Point | 250.42°C (rough estimate) |
| Flash Point | 137.1°C |
| Solubility | Chloroform, Dichloromethane |
| Vapor Presure | 0.000524mmHg at 25°C |
| Appearance | Solid |
| Color | White to beige |
| Storage Condition | Sealed in dry,Room Temperature |
| Refractive Index | 1.5769 (estimate) |
| MDL | MFCD00001852 |
Propanedinitrile, (1-ethoxyethylidene)- - Risk and Safety
| Risk Codes | R20/21/22 - Harmful by inhalation, in contact with skin and if swallowed. R36/37/38 - Irritating to eyes, respiratory system and skin. |
| Safety Description | S26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. S28 - After contact with skin, wash immediately with plenty of soap-suds. S36/37/39 - Wear suitable protective clothing, gloves and eye/face protection. S45 - In case of accident or if you feel unwell, seek medical advice immediately (show the label whenever possible.) |
| UN IDs | UN 3439 6.1/PG 3 |
| WGK Germany | 3 |
| RTECS | TY1510000 |
| HS Code | 29269090 |
| Hazard Class | 6.1(b) |
| Packing Group | III |
Propanedinitrile, (1-ethoxyethylidene)- - Reference Information
| Introduction | boiling point of 1-oxomaleonitrile: 312.6°C at 760mmHg, melting point: 90-92°C(lit.), it is a typical malononitrile derivative and is a common herbicide and growth regulator. |
| Application | 1-oxomaleonitrile exhibits good activity in pesticides, fungicides and other fields have in-depth research. |
| preparation | 4-pentylbenzoic acid methyl ester was prepared by alkaline hydrolysis, trans-4-pentylcyclohexanecarboxylic acid of high purity was prepared by hydrogenation of aromatic rings and isolated by crystallization. The performance of a ruthenium-containing catalyst in the hydrogenation of aromatic rings is more effective than that of a platinum or palladium-carbon catalytic system. In recent years, we have developed and applied highly efficient ruthenium nickel-carbon catalyst, RNC-5 (5% Ru - Ni/C, Ru:Ni = 9:1). The catalytic system can be synthesized in colleges and universities, and several different stereoisomers can be obtained to obtain trans 4-alkyl cyclohexyl carboxylic acid derivatives with high content. The scheme for the synthesis of trans-4-pentylcyclohexanecarboxylic acid is shown in the figure below. Fig.1 synthesis scheme of trans-4-pentylcyclohexanecarboxylic acid |
| EPA chemical substance information | information is provided by: ofmpeb.epa.gov (external link) |
Last Update:2024-04-09 21:04:16
Supplier List
Product Name: 1-(Ethoxycarbonyl)-3-cyclopentene-1-carboxylic acid Request for quotation
CAS: 5417-82-3
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 5417-82-3
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Product Name: ethoxyethylidene malononitrile Request for quotation
CAS: 5417-82-3
Tel: 13247110337
Email: 2205437118@qq.com
Mobile: 13247110337
QQ: 2205437118
Wechat: 13247110337
CAS: 5417-82-3
Tel: 13247110337
Email: 2205437118@qq.com
Mobile: 13247110337
QQ: 2205437118
Wechat: 13247110337
Spot supply
Product Name: (1-Ethoxyethylidene)malononitrile Visit Supplier Webpage Request for quotationCAS: 5417-82-3
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: (1-ETHOXYETHYLIDENE)MALONONITRILE Request for quotation
CAS: 5417-82-3
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 5417-82-3
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
Product Name: (1-Ethoxyethylidene)malononitrile Visit Supplier Webpage Request for quotation
CAS: 5417-82-3
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 5417-82-3
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Product Name: 1-(Ethoxycarbonyl)-3-cyclopentene-1-carboxylic acid Request for quotation
CAS: 5417-82-3
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 5417-82-3
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Product Name: ethoxyethylidene malononitrile Request for quotation
CAS: 5417-82-3
Tel: 13247110337
Email: 2205437118@qq.com
Mobile: 13247110337
QQ: 2205437118
Wechat: 13247110337
CAS: 5417-82-3
Tel: 13247110337
Email: 2205437118@qq.com
Mobile: 13247110337
QQ: 2205437118
Wechat: 13247110337
Spot supply
Product Name: (1-Ethoxyethylidene)malononitrile Visit Supplier Webpage Request for quotationCAS: 5417-82-3
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: (1-ETHOXYETHYLIDENE)MALONONITRILE Request for quotation
CAS: 5417-82-3
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 5417-82-3
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
Product Name: (1-Ethoxyethylidene)malononitrile Visit Supplier Webpage Request for quotation
CAS: 5417-82-3
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 5417-82-3
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History




